Share your photos by tagging us and using the hashtag #BigGreenEgg.
Want to see how the EGG is made? Click to Watch
Interesting Explanation of the Plateau
The Stall. The Zone. The Plateau. It has many names and it has
freaked out many a barbecue cook. I know because they email me right in
the middle of their cook. Panicky.
You get a big hunk-o-meat, like a pork shoulder for pulled pork or a beef brisket,
two of the best meats for low and slow smoke roasting, and you put it
on the smoker with dreams of succulent meat dancing in your head. You
insert your fancy new digital thermometer probe, stabilize the cooker at about 225°F and go cut the lawn. Then you take a nap.
The temp rises steadily for a couple of hours and then, to your
chagrin, it stops. It sticks. It stalls for four or more hours and
barely rises a notch. Sometimes it even drops a few degrees. You check
the batteries in your meat thermometer. You tap on the smoker
thermometer like Jack Lemon in the China Syndrome. Meanwhile the guests
are arriving, and the meat is nowhere near the 190°F mark at which these
two cuts are most tender and luscious. Your mate is tapping her foot
and you're pulling your hair out.
Sterling Ball of BigPoppaSmokers.com,
a major retailer of grills and smokers and a successful competition
cook says that "no matter what I tell customers, when the stall hits
them, they are horrified. It seems to last forever. They crank up the
heat. They bring the meat indoors and put it in the oven. They call me
at all hours."
What the heck is happening?
Many pitmasters have long believed that the stall was caused by a
protein in the meat called collagen combining with water and converting
to flavorful and slippery textured gelatin. Called a "phase change" the
conversion of collagen starts happening at about 160°F. Others have
speculated that the stall was the fat rendering, the process of lipids
turning liquid. Still others thought it was caused by protein
denaturing, the process of the long chain molecules breaking apart (for
more about these processes see my article on meat science).
But it is none of these things. It is much simpler, and there is a cure if you want it.
Dr. Blonder to the rescue
Dr. Greg Blonder,
is a physicist, entrepreneur, and former Chief Technical Advisor at
AT&T's legendary Bell Labs. He is also an avid cook, a barbecue
lover, and the resident myth-buster at AmazingRibs.com. He set out to figure out what causes the stall. His answer: "The stall is evaporative cooling."
It's that simple. The meat is sweating, and the moisture
evaporates and cools the meat just like sweat cools you after cutting
the lawn. Here's how he proved it.
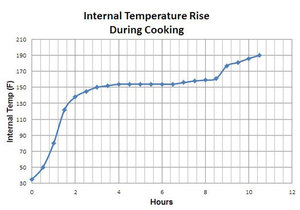
He charted a cook of a brisket on a thermostatically controlled
smoker. In his test (see the chart to the right) you can see the stall
starts after about 2 to 3 hours of cooking at about 150°F and then lasts
about 6 hours before the temp begins rising again. Your graph may vary
depending on the type of meat, its size, and your cooker, but the curve
should be similar.
Next he did some calculations and determined that the amount of
energy required to melt the collagen would be far less that that
consumed during the stall. A pork shoulder is about 65% water, 18%
protein, 15% fat, and 2% sugars and minerals. About 1/4 of the protein,
about 4% of the meat, is collagen.
Here's the logic: The fuel in your cooker (oxygen plus charcoal, gas,
or pellets) burns and produces energy which enters the cooking chamber
in the form of heat. Some of it escapes through the metal sides and some
goes out the vents, but some is absorbed by the cold meat. When it
heats, some of the energy is used up raising the temp of the entire
hunk, some of it is used in changing the chemistry and physical
structure of the molecules in the meat, and some is used to melt fat and
evaporate moisture. Pork shoulders and brisket have relatively high
connective tissue content. These connective tissues form a sheath around
muscle cells that connect them to each other, it sheaths bunches of
muscles into fibers, it encases fibers into whole muscles, and it
connects muscles to bone in the form of tendons and ligaments. Some are
made of really tough stuff called elastin. But some are made of
collagen. But the math didn't add up. There's just not enough collagen
to suck up all the energy necessary to prevent the meat from increasing
in temp. So it had to be something else, and his final test proved it.
 Hypothesizing
Hypothesizing
that the stall might be evaporative cooling, but still wondering if it
may be fat melting, Blonder took a lump of pure beef fat from the
fridge, inserted a thermometer probe, and placed it in a
thermostatically controlled smoker. He also soaked a large cellulose
sponge in water, shook it out, inserted a probe and placed it next to
the fat. Then he set the smoker for 225°F.
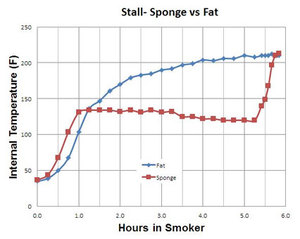
The results are pretty clear. The sponge is the red line and the fat is
the blue line. The fat did not have a stall at all. It slowly and
steadily heated on a nice gradual curve. But brother, did the sponge
ever stall. It climbed at about the same rate as the fat for the first
hour to about 140°F, and then it put on the breaks. In fact, it even
went down in temp! When it dried out after more than 4 hours, it took
off again.
The conclusion was inescapable: "Since there was a deep,
glistening pool of melted fat in the smoker, the rendering fat
hypothesis is busted. The barbecue stall is a simple consequence of
evaporative cooling by the meat's own moisture slowly released over
hours from within it's pores and cells. As the temperature of cold meat
rises, the evaporation rate increases until the cooling effect balances
the heat input. Then it stalls, until the last drop of available
moisture is gone."
Blonder points out that the size, shape, and surface texture of the
meat can influence the stall by influencing how much moisture is
available for evaporation, not to mention airflow and humidity in the
cooker. He also has done experiments that prove that the more airflow in
the oven the lower the stall temp so the amount of draft in your smoker
will impact the process. For example, pellet smokers, which have a fan
in them, create a convection environment and that speeds the
evaporation, so the stall can be shorter. I might add that ambient temp
and wind and other environmental factors also come into play.
The process seems to take all the moisture out from the surface and
just below it, and this is clearly part of the formation of the crusty,
jerky like, spice laden "bark" on the surface that contributes to the
textural and flavor profile. Why doesn't the meat just stay in the stall
until it is all dried out? "Much of the moisture in meat is tied up and
bound to other molecules like the collagen, fat, and protein. The
supply of moisture that can evaporate is limited. Once the meat's
ability to supply moisture peaks, it gradually starts to heat up."
Anyone who cooks large cuts knows that it is common for them to lose
as much as 25% of their weight during cooking. Well if you've ever
collected the drippings, you know that the melting fat is nowhere near
25%. The loss is mostly moisture. Considering that meats are 60 to 70%
water, that means there is still plenty of water left behind after
breaking out of the stall.
Will basting the meat or putting a water pan in the smoker impact the
stall? "There is no question extra humidity will slow down the cooking
process, whether it comes from a water pan or wet mop." When we baste,
whether by mopping, brushing, or spritzing, we cool the meat just by the
fact that the liquid is cool. It then sits on the surface and
evaporates prolonging the stall. When we put a water pan in the cooker,
the moisture evaporates from the surface and raises the humidity in the
cooker, slowing the evaporation from the meat, and slowing the cooking.
"In low and slow cooking this allows the meat's interior to catch up
with the surface temperature" explains Blonder.
Until now I had always believed that water pans were important to
keep the cooking chamber high humidity and thereby reduce moisture loss
from the meat. Apparently it does this somewhat, but they also cause the
cooking to take longer. But this is no reason to stop using water pans
because the moisture in the atmosphere inside the cooking chamber mixes
with the smoke, influences flavor, and lets the meat's interior catch up
with the exterior so it cooks more uniformly. Water pans also help
stabilize the temp in a charcoal fire because it heats and cools slowly
and this tends to even out spikes and valleys in a cooker.
Apparently the stall is not unique to barbecue. Blonder has proven it can happen in baked goods.
He points out that when we put ice cubes in a pan and turn on the heat,
the ice remains 32°F and the water from the melting ice remains close
to 32°F until all the ice is melted. This is a form of stall. Then the
water in the pan rises to 212°F, the boiling point, and stalls there
until the water is all gone, regardless of how much energy you apply to
the pan. Same phenomenon.
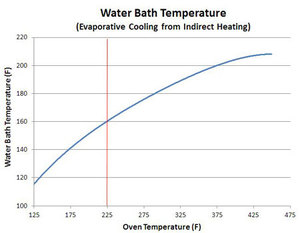 Interestingly,
Interestingly,
meat won't stall at high temps. Stalling is primarily a phenomenon of
low temp cooking. Blonder discovered this by putting a bowl of water in
an oven and set the thermostat for 125°F. The water stalled at about
115°F. Then he put another bowl in at 175°F. It stalled at 140°F. He
repeated the experiment in 50°F intervals. With each step, the stall
temp rose until it slowly approached the boiling point, 212°F, with the
oven just over 425°F. The bowl of water he cooked at 225°F stalled at
160°F. Well 225°F is the same temp of the oven in his other experiments,
not to mention the temp favored by most barbecue cooks, and 160°F is
pretty close to the stall temp for meat. That's the red line in the
chart at right.
When I showed this research to Ball he roared "I love it. It debunks
the urban legend that it is the collagen or fat melting. And it makes
great sense. This explains a lot! I can use this info!"
How to beat the stall and retain more moisture
How can we use this info? As you can see from the last chart, one way
to beat the stall and retain more moisture would be to cook at a higher
temp, and the fact is that more and more competition cooks are doing
just that. They figured it out by trial and error. Many now roast pork
shoulder in the 250°F range, and others are baking brisket north of
300°F.
There is a better way to prevent the stall, speed up cooking, and
retain moisture. For years, competition cooks have employed a trick
called the Texas Crutch. The crutch is a method of wrapping the meat
with aluminum foil and adding a splash of liquid like apple juice or
beer. The conventional wisdom was that the moisture created a bit of
steam that tenderized the meat, and since steam conducts heat faster
than air, it speeds cooking. Typically they do the wrapping when the
meat hit 170°F or so, deep into the stall.
Blonder says that there is no steaming action inside the foil at
225°F. Foil prevents evaporative cooling and over a period of hours the
temperature inside the foil slowly approaches a low simmer. Any moisture
that comes out of the meat just pools in the foil along with the liquid
the cook adds. "It's like running a marathon in a rain coat. You'll
sweat, but it won't cool you off." There is a slight fog inside the
foil, but no steam, so there is no steam cooking. But there is a form of
braising! Braising is a wet method of cooking similar to stewing or
poaching but the food is usually not submerged as they are in those
methods. It is more like what happens in a slow cooker.
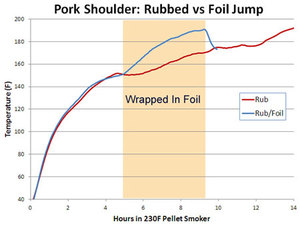 For
For
his final test, Blonder took a 6 pound pork butt and divided it in two
removing the bone. He rubbed them both with a standard pork rub and put
them into a 230°F cooker until the stall began. Then he wrapped one in
foil and added 1 tablespoon of water. In the chart at right, it is the
blue line, labeled "Rub/foil".
The other he left alone, naked except the rub, the red line labeled
"Rub". As you can see the wrapped pork climbed to 180°F in about half
the time, about 6 hours. He let it go to 190°F, a target I recommend,
removed the foil and put it back on to firm up the bark. As you can see,
the temp dropped immediately after unwrapping as the moisture
evaporated and cooled the meat. After 4 hours the unwrapped butt had
still not passed 180°F. The lines end when he got hungry and when the
foiled/unfoiled butt hit the same temp as the never foiled butt. He
called the foiled butt "Really juicy and nearly perfect." But "When the
other hit 180°F the meat was still slightly tough. It needed another
hour or so to finish cooking in kitchen oven."
At right are photos of the two pieces of pork. Pretty comparable.
If the stall was caused by conversion of collagen to gelatin, since
the transition happens within the foil and there is no stall, the phase
change of collagen cannot be the cause. The fact that collagen melts at
about the same temp as the stall is a coincidence, not the cause of the
stall.
Based on Blonder's data, I now recommend that you wrap pork shoulders
and beef briskets at about 150°F, after about 2 hours in the smoke. By
then it has absorbed as much smoke as is needed. If you wrap it then,
the meat powers right through the stall on a steady curve and takes much
less time. It also retains more juice.
Ball says that he is now following a similar protocol in competition.
He won't say what temp he cooks at on his MAK pellet smoker, but he is
now foiling when his bark is the deep mahogany color he wants, usually
somewhere between 140 and 150°F. He leaves it in the foil all the way up
to the end, takes it out of the cooker, lets it come down in temp to
about 175°F so it stops cooking, and then wraps it in a towel and puts
it in an insulated holding box called a cambro for an hour or two to
rest (see my article on how you can rig a faux cambro).
There is a problem with this approach for some cooks: The meat does
not have a hard chewy bark on the exterior. Ball says that a hard bark
is emblematic of overcooked meat. He wants a dark, flavorful, tender
bark. But if you want a hard bark, the solution is to pull the meat out
of the foil when it hits 180°F or so, and hit it with higher heat to dry
the exterior and darken the rub. Or just skip the foil altogether, do
things the old fashioned tried and true way, and just be patient. Either
way, the results are superb.
If you change the way you cook based on Blonder's work, let us know how you liked the outcome. Click here to go to Blonder's website for more details on his experiments with the stall.
Comments
-
Nice post. Post of the day nominee from me. So much for the collagen theory.
-
Bingo!!!
-
Is Cotto202 Meathead?
-
I'm going to do a turbo packer this weekend to give this a run. I'm an old school low and slow guy and had fully bought in to and defended the collagen theory until reading this a few weeks back. I'll never stop going low and slow but it would be awesome to do a brisket in 4-6 hours! We'll see. I'll report back Sunday PM when it's done.Keepin' It Weird in The ATX FBTX
-
I'm going to do a turbo packer this weekend to give this a run. I'm an old school low and slow guy and had fully bought in to and defended the collagen theory until reading this a few weeks back. I'll never stop going low and slow but it would be awesome to do a brisket in 4-6 hours! We'll see. I'll report back Sunday PM when it's done.
And think of the $$$ you will save with the reduced beer consumption time-looking forward to the results.
Louisville; Rolling smoke in the neighbourhood. # 38 for the win. Life is too short for light/lite beer! Seems I'm livin in a transitional period. -
I knew you had to be lurking close by on this one. I have been known to drink beer before AND after the brisket is in the egg so I'm not sure a reduction in consumption is in the cards.I'm going to do a turbo packer this weekend to give this a run. I'm an old school low and slow guy and had fully bought in to and defended the collagen theory until reading this a few weeks back. I'll never stop going low and slow but it would be awesome to do a brisket in 4-6 hours! We'll see. I'll report back Sunday PM when it's done.
And think of the $$$ you will save with the reduced beer consumption time-looking forward to the results.Keepin' It Weird in The ATX FBTX -
@Cen-Tex-Not surprised by that approach at all-makes sense to me. Would be good to have the high-speed cook option when time is short...Louisville; Rolling smoke in the neighbourhood. # 38 for the win. Life is too short for light/lite beer! Seems I'm livin in a transitional period.
-
@Cen-Tex-Not surprised by that approach at all-makes sense to me. Would be good to have the high-speed cook option when time is short...
I'm all for trying new things and it wouldn't hurt my feelings if people thought I stayed up all night carefully crafting their brisket Keepin' It Weird in The ATX FBTX
Keepin' It Weird in The ATX FBTX -
I'm going to do a turbo packer this weekend to give this a run. I'm an old school low and slow guy and had fully bought in to and defended the collagen theory until reading this a few weeks back. I'll never stop going low and slow but it would be awesome to do a brisket in 4-6 hours! We'll see. I'll report back Sunday PM when it's done.
Lot of people going over to turbo recently. Really interested to se the results. Briskets are notoriously tough here and they are always hit and miss for me
Steve
Caledon, ON
-
Great post.
-
Thanks for posting this. I had read that article a while back and couldn't remember where I saw it or the finer points of it. Nice find.Sarasota, FL via Boynton Beach, FL, via Sarasota, FL, via Charleston, SC, via The Outer Banks, via God's Country (East TN on Ft. Loudon Lake)
-
Awesome read. As someone who is new to grilling its good to moe in with quality info like this.Boom
-
Great read...
-
I have foiled the last two butts that I did to get them done on time. They were both great. I really see no reason not to do them this way. Saves time and fuel and comes out just as good if not better.
George
George -
This is a great article especially for newbie's like myself to smoking. This article should be included with every new BGE. Thanks Doc!
-
Dang! I feel much smarter than I did just a few minutes ago!
Thanks for sharing.
Gittin' there... -
Thanks for posting Cotto very informative._________________________________________________Don't let the truth get in the way of a good story!Large BGE 2006, Mini Max 2014, 36" Blackstone, Anova Sous Vide
Green Man GroupJohns Creek, Georgia -
I agree with Fearless. I am a better person now. Hard to argue with science - especially when it makes things easier and taste better!
2 LBGE, 1 SBGE, Stoker Wifi, White Thermapen, Igrill---Gilbert SC
Categories
- All Categories
- 182.7K EggHead Forum
- 15.7K Forum List
- 459 EGGtoberfest
- 1.9K Forum Feedback
- 10.3K Off Topic
- 2.2K EGG Table Forum
- 1 Rules & Disclaimer
- 9K Cookbook
- 12 Valentines Day
- 91 Holiday Recipes
- 223 Appetizers
- 516 Baking
- 2.4K Beef
- 88 Desserts
- 163 Lamb
- 2.4K Pork
- 1.5K Poultry
- 30 Salads and Dressings
- 320 Sauces, Rubs, Marinades
- 543 Seafood
- 175 Sides
- 121 Soups, Stews, Chilis
- 35 Vegetarian
- 100 Vegetables
- 313 Health
- 293 Weight Loss Forum